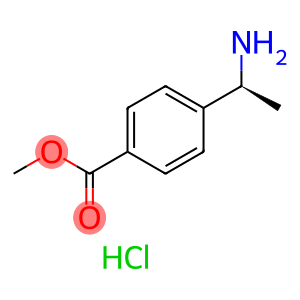
methyl (S)-4-(1-aminoethyl)benzoate hydrochloride
(S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride
CAS: 847728-91-0
Molecular Formula: C10H14ClNO2
methyl (S)-4-(1-aminoethyl)benzoate hydrochloride - Names and Identifiers
methyl (S)-4-(1-aminoethyl)benzoate hydrochloride - Physico-chemical Properties
| Molecular Formula | C10H14ClNO2 |
| Molar Mass | 215.68 |
| Storage Condition | Sealed in dry,Room Temperature |
methyl (S)-4-(1-aminoethyl)benzoate hydrochloride - Introduction
(S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride is an organic compound with the chemical formula C10H14ClNO2. The following is its nature, use, preparation and safety information:
Nature:
-Appearance: It is a white crystalline solid or crystalline powder form.
-Solubility: It is soluble in water and organic solvents, such as ethanol and ether.
-Melting point: Its melting point is usually in the range of 160-164°C.
-Optical activity: Due to the chirality of its molecular structure, the compound is an optically active substance with a levorotatory (S) configuration.
Use:
- (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride is an important intermediate compound, which is widely used in organic synthesis reactions.
-It is commonly used in the synthesis of drugs and biologically active compounds, such as anti-tumor drugs, antidepressants and neurotransmitter mimics.
Preparation Method:
(S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride has many preparation methods. A common method is:
1. the reaction of methyl benzoate with ammonia ethanol under suitable reaction conditions to generate N-benzyl ethanol benzamide.
2. reacting N-benzylethanolbenzamide with hydrochloric acid to obtain (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride.
Safety Information:
- (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride have low toxicity when properly used and stored.
-During the operation, the laboratory safety operation procedures should be observed.
-Avoid contact with eyes, skin and respiratory tract, and avoid inhaling dust.
-For the use of this compound, please refer to the relevant Chemical Safety Data Sheet (MSDS) for more detailed safety information.
Nature:
-Appearance: It is a white crystalline solid or crystalline powder form.
-Solubility: It is soluble in water and organic solvents, such as ethanol and ether.
-Melting point: Its melting point is usually in the range of 160-164°C.
-Optical activity: Due to the chirality of its molecular structure, the compound is an optically active substance with a levorotatory (S) configuration.
Use:
- (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride is an important intermediate compound, which is widely used in organic synthesis reactions.
-It is commonly used in the synthesis of drugs and biologically active compounds, such as anti-tumor drugs, antidepressants and neurotransmitter mimics.
Preparation Method:
(S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride has many preparation methods. A common method is:
1. the reaction of methyl benzoate with ammonia ethanol under suitable reaction conditions to generate N-benzyl ethanol benzamide.
2. reacting N-benzylethanolbenzamide with hydrochloric acid to obtain (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride.
Safety Information:
- (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride have low toxicity when properly used and stored.
-During the operation, the laboratory safety operation procedures should be observed.
-Avoid contact with eyes, skin and respiratory tract, and avoid inhaling dust.
-For the use of this compound, please refer to the relevant Chemical Safety Data Sheet (MSDS) for more detailed safety information.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride Visit Supplier Webpage Request for quotationCAS: 847728-91-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-4-(1-AMINO-ETHYL)-BENZOIC ACID METHYL ESTER HYDROCHLORIDE Request for quotation
CAS: 847728-91-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 847728-91-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (S)-4-(1-AMINO-ETHYL)-BENZOIC ACID METHYL ESTER HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 847728-91-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 847728-91-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-Methyl 4-(1-aminoethyl)benzoate hydrochloride Visit Supplier Webpage Request for quotationCAS: 847728-91-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (S)-4-(1-AMINO-ETHYL)-BENZOIC ACID METHYL ESTER HYDROCHLORIDE Request for quotation
CAS: 847728-91-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 847728-91-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (S)-4-(1-AMINO-ETHYL)-BENZOIC ACID METHYL ESTER HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 847728-91-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 847728-91-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
methyl (S)-4-(1-aminoethyl)benzoate hydrochloride
2594907-89-6
135-49-9
36852-02-5
弱酸性橙 PR
METHYL TRICHLOROACETATE 1X1ML MTBE 10&
1-溴-6H-二苯并[B,D]吡喃
119580-41-5
2,5-二氢-2,5-二甲氧基呋喃, 顺反异构体混合物
2594907-89-6
135-49-9
36852-02-5
弱酸性橙 PR
METHYL TRICHLOROACETATE 1X1ML MTBE 10&
1-溴-6H-二苯并[B,D]吡喃
119580-41-5
2,5-二氢-2,5-二甲氧基呋喃, 顺反异构体混合物


